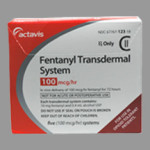
The FDA (Food and Drug Administration) is reminding patients who use “fentanyl transdermal system pain patches (Duragesic and its generics) to always store, use, and dispose of them carefully to prevent unintended poisonings and other harm, especially to children and pets.” Fentanyl should only be handled by the patient or the patient’s caregivers. Fentanyl is [...]